Contact lenses recalled over fears they could cause ‘eye redness, discomfort or corneal abrasion’
27 September 2019, 17:05
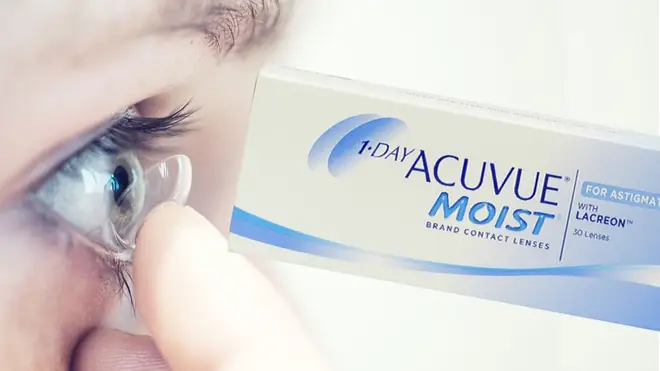
Johnson & Johnson’s are recalling batches of their contact lenses after receiving reports of a “foreign matter” on the lens or in the solution.
Customers who have purchased 1-day Acuvue Moist for Astigmatism lenses by Johnson & Johnson’s are being told to return certain batches to suppliers.
The Medicine and Healthcare products Regulatory Agency (MHRA) are recalling some of the products over fears there are dangerous particles on the lens or in the solution.
Customers are being asked to check the batch number on their boxes of 1-day Acuvue Moist for Astigmatism lenses to see if they need to return the product.
You can find the list of affected batch numbers here.
This comes after the company received reports of a “foreign matter on the contact lens or in the contact lens blister solution”.
They say there has only been one report of someone suffering discomfort and eye redness from the lens, and they add that there have been “no reports of serious adverse events”.
The company explained: “We received a limited number of reports of foreign matter on the contact lens or in the contact lens blister solution. While there has been one report of lens use that resulted in discomfort and eye redness, importantly, there have been no reports of serious adverse events.
“Based on a safety review by our Medical team, the presence of these small particles is associated with low potential risk if a patient inserts an affected lens in their eye. If the particles weren’t noticed before insertion in the eye, it could result in eye redness, discomfort, or corneal abrasion.”

They added: “We have identified the cause, taken corrective action, and are planning to implement even stronger manufacturing and quality controls based on learnings from this event.
MHRA group manager, device safety and surveillance, Mark Birse said: “Whilst the risk is low, the MHRA takes the safety of the medicines and devices we regulate very seriously.
“It is therefore important that as many customers as possible are made aware of the need to check their lenses for these lot numbers.”


































